
Test results are available approximately 15 minutes after a plasma sample is inserted in the test i-STAT cartridge. Abbott's i-STAT TBI Plasma test is the first rapid test on a portable analyzer to receive FDA 510(k) clearance and can help determine the need for a computed tomography (CT) scan. I hope the release of this preliminary data will prompt other clinical laboratories to review their results. They can be measured with diagnostic testing. 7 days of the scan or the patient has not had this test. I have decided to discontinue bedside INR testing with ISTAT until this discrepancy has been resolved. The following patients will require a POC creatinine tests if the recent test result is not within. This small single study suggests that clinicians cannot make accurate clinical decisions based on I-STAT INR results. ISTAT INR was as much as 0.5 lower than STAR when the STAT INR is 2.8.The secondary objective of this study is the collection of additional data and. Single-use i-STAT test cartridges cover a broad menu of testing options in a. The purpose of this study is to evaluate the clinical performance of the i-STAT TBI test for the proposed intended use to assist in determining the need for a CT scan in patients presenting with suspected mild traumatic brain injury who are 18 years of age or older. This study demonstrated significant INR differences between I-STAT and STAR. These results were much more discrepant than the results published in the I-STAT Prothrombin Time/PT packgage insert that compared I-STAT to STA Compact. Even more disconcerting was that the bias was inconsistent throughout the therapeutic range. Test with the patient in minutes no delays caused by transporting samples. Recently, I compared the INR results of patients treated with warfarin who were simultaneously tested with a fingerstick specimen on an I-STAT at the bedside and a venipuncture specimen on a Stago STAR coagulation analyzer located in the main clinical laboratory. This revolutionary, handheld analyzer provides reliable test results in just minutes, using as little as two drops of blood and a test cartridge. I-STAT INR cartridges are used to determine if a patient presenting to the ED with an ischemic stroke can receive tissue plasminogen activator or if a patient anticoagulated with warfarin can undergo an emergent invasive procedure. Results of Experiments on Big Data, characterised, by their very nature, by the use of non-official sources.Įxperimental statistics are available in chronological order also.I-STAT (Abbott Point of Care) is a popular device for point of care testing that is used in hospitals to provide quicker turnaround time and expedite clinical decision making.Interpretation frameworks and analysis of complex phenomena obtained through the integration of official sources.New indicators produced through the integration of a multiplicity of official and non-official sources in this case, the focus is on phenomena under investigation rather than on statistical sources used to describe them.Non-standard classifications produced on the basis of the official taxonomies defined at an international level and currently used by Istat, or proposed as experimental within analysis and research activities based on microdata processing The i-STAT measures multiple pathology test profiles including blood electrolytes, urea, creatinine, glucose, blood gases, lactate, cardiac troponin I and.To ease users in finding and utilising them, experimental statistics produced by Istat are organised in four different typologies: For the glucose test strip and other cartridge tests, test sample within 30 minutes of collection. They are experimental statistics because they do not respect all the steps necessary to test new methodologies, to transform them into technological and organisational solutions, to verify if quality requirements and harmonisation rules are fulfilled.īut their potential is really high they fill knowledge gaps in a timely way they serve as a driving force for new analyses and indicators they guarantee an important information support to policies. The results are made available for users’ use and evaluation. 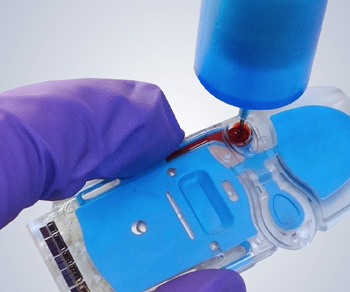
In line with Eurostat and other National Statistical Institutes, Istat experiments with the use of new sources and the application of innovative methods in producing data. Laboratory for Elementary Data Analysis.Quality in the National statistical system.Public administrations and private institutions.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
